 Main group elements are sometimes called representative elements. Note that down each group, the configuration is often similar. The colored sections of Figure 1 show the three categories of elements classified by the orbitals being filled.įigure 1: This version of the periodic table shows the configuration of each element. Now the arrangement of the periodic table is understood elements whose atoms have the same number of valence electrons are in the same group. It is important to remember that the periodic table was developed on the basis of the chemical behavior of the elements, well before any idea of their atomic structure was available. The similarity in chemical properties among elements of the same group occurs because they have the same number of valence electrons. It is the loss, gain, or sharing of valence electrons that defines how elements react. Elements in any one group (or column) have the same number of valence electrons (Figure 1) the alkali metals lithium and sodium each have only one valence electron, the alkaline earth metals beryllium and magnesium each have two, and the halogens fluorine and chlorine each have seven valence electrons. Across a period, each consecutive element has an additional proton in the nucleus and an additional electron to the valence shell. The horizontal rows are known as periods. Valence electrons are also the determining factor in some physical properties of the elements. The outer electrons have the highest energy of the electrons in an atom and are more easily lost or shared than the core electrons. 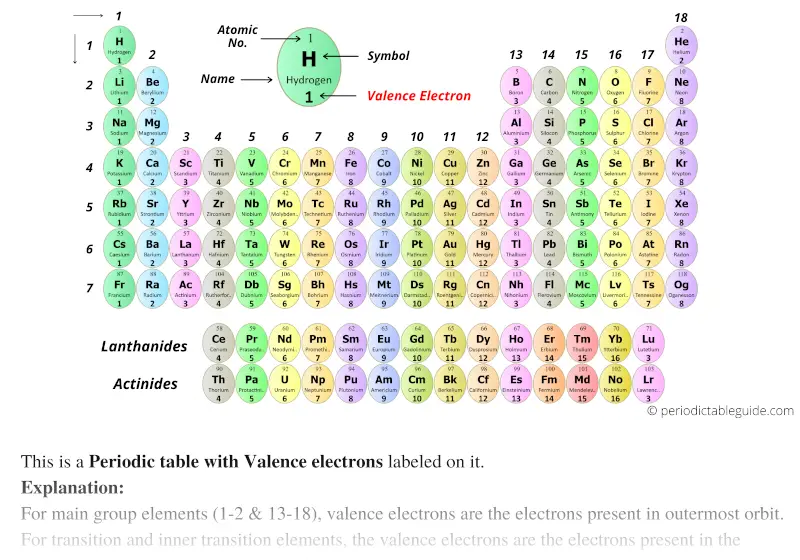
Because they are in the outer shells of an atom, valence electrons play the most important role in chemical reactions. 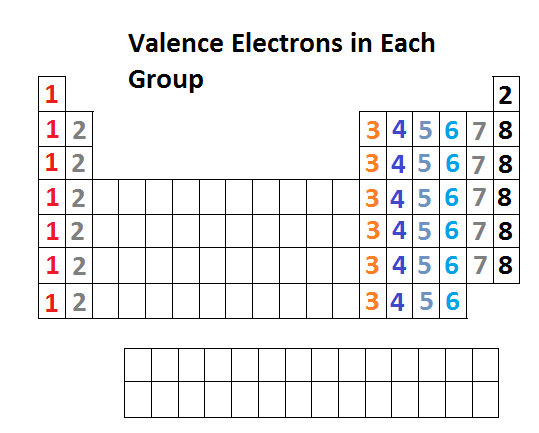 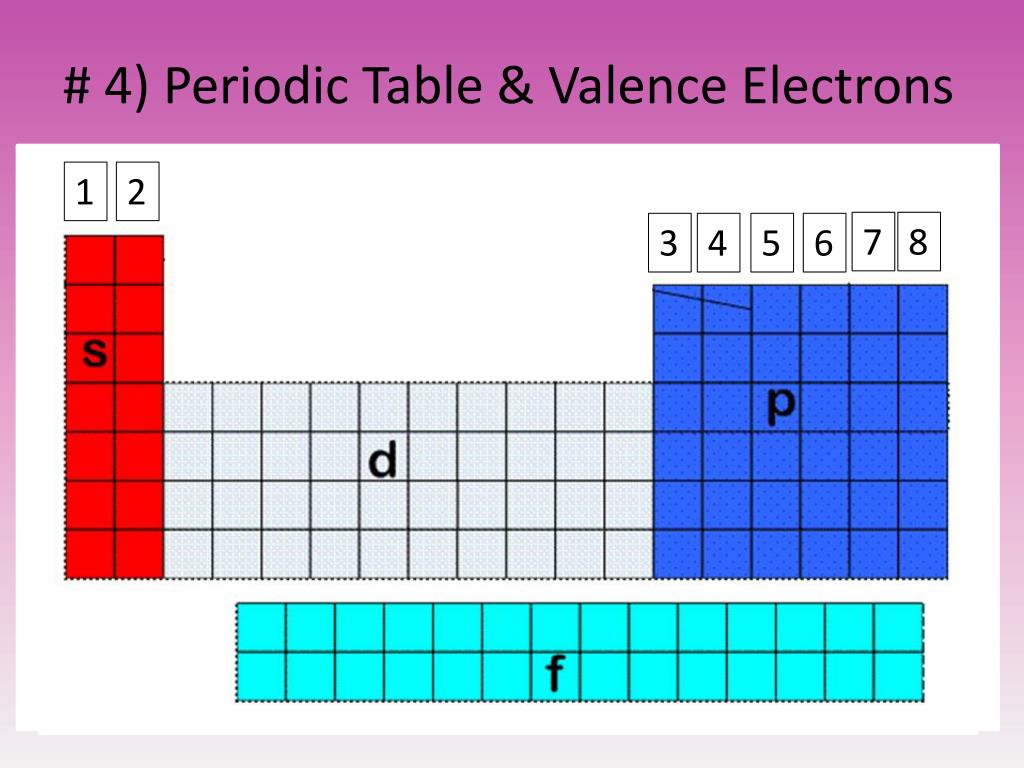
When their electron configurations are added to the table, a periodic recurrence of similar electron configurations in the outer shells of these elements is observed. The periodic table arranges atoms based on increasing atomic number so that elements with the same chemical properties recur periodically. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
